Executive Summary
The chemical process of changing seawater into potable or fresh water is called desalination. Thermal distillation and membrane processes are the two main approaches used around the world to desalinise water. Desalination processes may be used in municipal, industrial, or commercial applications. With improvements in technology, desalination processes are becoming cost-competitive with other methods of producing usable water to respond to a growing demand. Stand-alone desalination plants can use renewable energy to operate. The pure water that is obtained after desalination must be re-mineralised to be adequate for human consumption. The concentrated brine produced in desalination processes needs to be disposed of properly.
| In | Out |
|---|---|
Freshwater, Drinking Water, Non-biodegradable Wastewater |
Introduction
As populations increase and sources of high quality, fresh drinking water decrease, using desalination processes to provide freshwater when other sources and treatment procedures are uneconomical or not environmentally responsible is becoming more and more common.
Desalination is any process that removes excess salts and other minerals from water. In most desalination processes, saltwater (also called “feed water”) is treated and two streams of water are produced:
- Treated freshwater that has low concentrations of salts and minerals.
- Concentrate or brine, which has salt and mineral concentrations higher than that of the feed water.

The feed water for desalination processes can be seawater or brackish water. Brackish water contains more salt than fresh water but less than saltwater. Brackish water is commonly found in estuaries, which are the lower courses of rivers where they meet the sea, and aquifers, which are stores of water underground.
Two technologies are primarily used around the world for desalination: thermal distillation and membranes. Both technologies need energy to operate and produce freshwater, and are described as follow:
- Thermal distillation: These technologies are widely used in the Middle East, primarily because the region’s petroleum reserves keep energy costs low. The three major, large-scale thermal processes are multistage flash distillation, multi-effect distillation, and vapour compression distillation. Another thermal method, solar distillation, is typically used for very small production rates.
- Membrane technologies: These are primarily used in the United States. These systems treat the feed water by using a pressure gradient to force the water through membranes. The three major membrane processes are electrodialysis, electrodialysis reversal, and reverse osmosis. Membrane distillation is an emerging membrane-based desalination technology.
Thermal technologies
Multi-stage flash distillation
Multi-stage flash distillation is a process that sends the saline feed water through multiple chambers. In these chambers, the water is heated and compressed to a high temperature and high pressure. As the water progressively passes through the chambers, the pressure is reduced, causing the water to rapidly boil. This boiling causes vapour to be produced in each chamber. The vapour, which is composed of freshwater, is then is condensed and collected.
Multi-effect distillation
Multi-effect distillation employs the same principals as the multi-stage flash distillation process except that instead of using multiple chambers of a single vessel (also known as an“effect”), multi-effect distillation uses successive vessels.
A series of evaporator vessels produces water at progressively lower pressures. Water boils at lower temperatures as pressure decreases, so the water vapour of the first vessel serves as the heating medium for the second, and so on. The more vessels there are, the higher the performance ratio will be.
The water vapour that is formed when the water boils is condensed and collected. The use of multiple vessels makes the multi-effect distillation process more efficient.
Vapour compression distillation
Vapour compression distillation can function independently or be used in combination with another thermal distillation process. Vapour compression distillation uses heat from the compression of vapour to evaporate the feed water. Vapour compression distillation units are commonly used to produce freshwater for small- to medium-scale purposes such as resorts, industries, and petroleum drilling sites.
Solar distillation
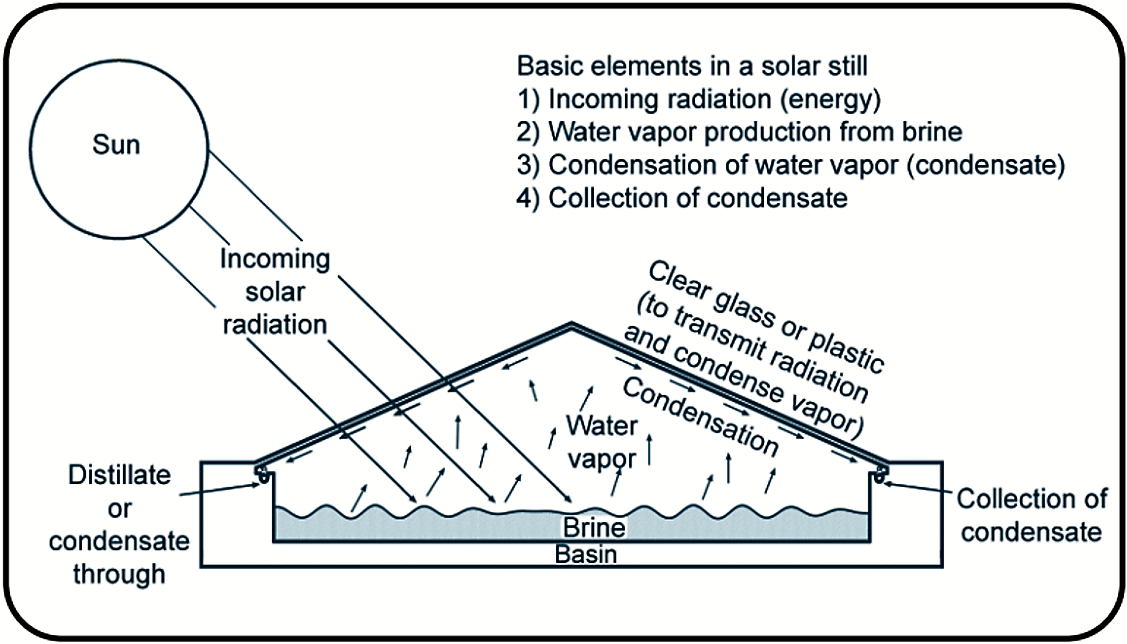
Solar desalination is generally used for small-scale operations. Although the designs of solar distillation units vary greatly, the basic principle is the same: that the sun provides energy to evaporate freshwater from saline water. In solar distillation, the water vapour formed from the evaporation process condenses on a clear glass or plastic covering and is collected as freshwater in a condensate trough. The covering is used to both transmit radiant energy and allow water vapour to condense on its interior surface. The salt and un-evaporated water left behind in the still basin form the brine solution that must be discarded appropriately.
Solar distillation is often used in arid regions where safe freshwater is not available. Solar distillation units produce differing amounts of freshwater, according to their design and geographic location.
Recent tests on four solar still designs by the Texas AgriLife Extension Service in College Station, Texas, have shown that a solar still with as little as 0.7 square meter surface area can produce enough water for a person to survive.
Membrane technologies
Electrodialysis and electrodialysis reversal
The membranes used in electrodialysis are built to allow the passage of either positively or negatively charged ions, but not both. Common ionic molecules in saline water are sodium, chloride, calcium, and carbonate. Electrodialysis uses the driving force of an electrical potential to attract and move different cations (positively charged ions) or anions (negatively charged ions) through the permeable membrane. The cations are attracted to the negative electrode, and the anions are attracted to the positive electrode. When the membranes are placed so that some allow only cations to pass and others allow only anions to pass, the process can effectively remove most of the salts from the water.
The electrodialysis reversal process functions like the electrodialysis process; the only difference is that in the reverse process, the charge of the electrodes is switched periodically. This reversal in flow of ions helps remove scaling and other debris from the membranes, which extends the system’s operating life.
Reverse osmosis
Reverse osmosis (RO) uses a pressure gradient as the driving force to move high-pressure saline feed water through a membrane that prevents the salt ions from passing (see also membrane filtration).
Because the RO membrane has such small pores, the feed water must be pre-treated adequately before being passed through it. The water can be pre-treated chemically, to prevent biological growth and scaling, and physically, to remove any suspended solids.

Membrane distillation
Adapted from GRYTA (2011)
Membrane distillation is a new industrial process where feed water is heated to increase its vapour pressure, which generates a difference between the partial pressure at both sides of the membrane. Hot water vapour passes through non-wetted pores of the hydrophobic membranes, which cannot be wetted by liquid water. The passing vapour is then condensed on a cooler surface to produce fresh water. In the case of solutions containing non-volatile substances, only water vapour is transported through the membrane. Thus, the process has a potential application for water desalination and wastewater treatment. Membrane distillation has significantly lower requirements concerning the pre-treatment of feed water; therefore, it enables the production of pure water from raw water sources. However, the formation of deposits both pollutes membrane surfaces and makes it easier for water to penetrate membrane pores. Consequently, the membranes lose their separation properties and the membrane distillation process stops. This is why it is essential to prevent formation of deposits on the membrane surfaces.
In membrane distillation, heat is required to evaporate the feed components (similar to classical distillation). However, since it operates at low temperatures (50-90°C), the energy demands for heating are relatively low, making it possible to use renewable energy sources to heat the feed water.
Alternative energy sources
Adapted from MATIOULAKIS et al. (2007)
Renewable energies and desalination plants are two different technologies that can be combined in various ways. The interface between the renewable energy systems and the desalination systems is where the energy generated by renewable energy is directed to the desalination plant. This energy can be in different forms, such as thermal energy, electricity or shaft power.
Renewable energy-driven desalination systems fall into two categories: thermal processes and electromechanical processes. Most desalination plants powered by renewable energy are stand-alone systems in locations without an electricity grid.
Stand-alone-systems are often hybrid systems, combining more than one type of renewable energy source, such as wind and solar energy, or including a diesel generator. In order to ensure continuous or semi-continuous operation independent of weather conditions, stand-alone systems usually include a storage system.
Mineralisation of water after desalination
Adapted from HASSON & BENDRIHEM (2006)
The lack of dissolved minerals in the high-purity water produced by desalination processes raises some problems. High-purity water tends to be highly reactive and, unless treated, it can create severe corrosion difficulties during its transport in conventional pipelines. Also, untreated desalinated water cannot be used directly as a source of drinking water. A certain degree of remineralisation is necessary in order to make the water palatable and for re-introducing some essential ions required for health considerations.
|
| Total dissolved solids | Calcium | Magnesium | Sodium | Chlorine | Sulphate |
| mg/L | 400 | 30-150 | 0-100 | 30-150 | 0-200 | 0-200 |
Minerals contained in “normal” water. Source: HASSON & BENDRIHEM (2006)
The most common processes for the remineralisation of desalinated water are as follows:
- Dosage of chemical solutions based on calcium chloride and sodium bicarbonate
- Lime dissolution by carbon dioxide
- Limestone dissolution by carbon dioxide. This is the simplest and most widely used process.
Operation and maintenance
Desalination plants’ operation and maintenance depends on the technology used. For instance, multi-stage flash distillations and reverse osmosis require trained technicians to operate and maintain the plants, whereas solar desalination requires very low maintenance.
Brine management
Both thermal and membrane desalination processes produce a stream of brine water that has a high concentration of salt and other minerals or chemicals that were either removed during the desalination process or added to help pre-treat the feed water. For all of the processes, the brine must be disposed of in an economical and environmentally friendly way.
Options for discharging the brine include discharge into the ocean, injection through a well into a saline aquifer, or evaporation. Each option has advantages and disadvantages. In all cases, the brine water should have a minimal impact on the surrounding water bodies or aquifers. Specific considerations for the water quality include saline concentration, water temperature, dissolved oxygen concentrations, and any constituents added as pre-treatment.
Costs considerations
Adapted from ZHOU & TOF (2004)
The costs of desalination vary significantly depending on the size and type of the desalination plant, the source and quality of incoming feed water, the plant location, site conditions, qualified labour, energy costs and plant lifetime. Lower feed water salinity requires less power consumption and dosing of chemicals to prevent scaling. Larger plant capacity reduces the unit cost of water due to economies of scale. Lower energy costs and a longer plant running period reduce unit product water cost.
The primary elements of desalination costs are capital cost and annual running cost. The capital cost includes the purchase cost of major equipment, auxiliary equipment, land, construction, management overheads, and contingency costs. The capital costs for seawater desalination plants have decreased over the years due to the on-going development of processes, components and materials. Annual running costs consist of costs for energy, labour, chemicals, consumables and spare parts. The energy costs strongly affect the running costs for thermal processes. Thus, distillation costs fluctuate more than reverse osmosis as they depend on changing energy costs. In regions where energy is fairly expensive, reverse osmosis is a favourable choice compared to any other thermal processes due to its lower energy consumption.
As with all new technologies, progress in desalinating water has been rapid. Whereas it cost about $9.0/m3 to desalinate seawater around 1960, the costs are now around $1.0/m3 for the multi-stage flash distillation process. For reverse osmosis, the most popular method, the costs have fallen to $0.6/m3 for brackish water desalination. There is no reason to believe that the trend will not continue in the future.
However, it should be noted that the costs of desalination still remain higher than other alternatives for most regions of the world.
At a glance
| Working principle | Removes salts from sea or brackish water to produce freshwater. Thermal technologies and membranes are the two most widely used techniques. |
| Capacity/adequacy | Technologies available are adapted to many scales from small to large communities. |
| Performance | Very efficient to produce deionised water. |
| Costs | Relatively high costs but are tending to decrease. |
| Self-help compatibility | Engineers are required for the design and trained technicians for the operation in many cases. |
| O&M | Depends on the process used. |
| Reliability | Highly reliable if plant is properly maintained. |
| Main strength | Can produce drinking water from very abundant source (oceans). |
| Main weakness | High energy consumption. |
Desalination processes are adapted to produce drinking water in areas where only seawater or brackish water is available. A number of technologies have been developed for desalination and can be used at different scales from small communities’ water supply (e.g. solar distillation) to huge plants (e.g. reverse osmosis) for cities.
Desalination costs, although continuously decreasing, are still higher than conventional drinking water supply processes. Consequently, desalination should be applied if it is the only option.
Pure water produced by desalination processes is corrosive and not appropriate for human consumption. Thus, remineralisation (e.g. limestone dissolution) is required before distribution and consumption.
A Review of Current Knowledge Desalination for Water Supply
Water Desalination by Membrane Distillation
Introduction to Desalination Technologies
Desalination by using Alternative Energy: Review of State of the Art
This paper presents a review of the highlights that have been achieved during the recent years and the state-of-the-art for most important efforts in the field of desalination by renewable energy, with emphasis on solar energy applications.
MATIOULAKIS, E. ; BELESSIOTIS, V. ; DELYANNIS, E. (2007): Desalination by using Alternative Energy: Review of State of the Art. In: Elsevier Desalination : Volume 203 , 346-365. URL [Accessed: 07.03.2019] PDFDesalination Methods for Producing Drinking Water
Desalination Units Powered by Renewable Energy Systems
This paper reports the proceedings of the Autonomous Desalination Units Based on Renewable Energy (ADU-RES) International Seminar held in Hammaet, Tunisia, focusing on the water supply situation in Tunisia and analysing different solutions based on desalination.
ADU-RES (2005): Desalination Units Powered by Renewable Energy Systems. Hammamet: ADU-RES URL [Accessed: 04.07.2012]The Economics and Performance of Desalination Plants
In this paper, the technical and economic features of the major seawater desalination processes are reviewed. Each process is described in some detail and the basic mathematical relations used for estimating its performance are outlined. Economic models are also introduced which were developed for the purpose of estimating the water cost for each process.
EL-NASHAR, A.M. (n.y): The Economics and Performance of Desalination Plants. Paris: Encyclopedia of Life Support Systems (EOLSS) URL [Accessed: 05.03.2012]Assessment of most Promising Developments in Solar Desalination
Indirect solar desalination systems consist of a conventional desalination unit coupled to a solar conversion system. Their most significant possibilities of development in the near future are assessed in this paper. Particular emphasis is given to the efficiency of such systems since their cost mainly depends on the maturity of the technology and the production scale.
GARCIA-RODRIGUEZ, L. (2007): Assessment of most Promising Developments in Solar Desalination. In: RIZZUTI ; (2007): Solar Desalination for the 21th Century. Heidelberg: 355-369. URL [Accessed: 07.03.2019] PDFSolar thermal-driven desalination plants based on membrane distillation
This paper describes the use of solar thermally driven stand-alone desalination systems adapted to remote areas based on membrane distillation. The aim is to develop systems for a capacity ranging from 0.2 to 20 m3/day. Technical simplicity, long maintenance-free operation periods and high quality output are the important parameters that will enable successful application of the systems.
KOSCHIKOWSKI, J. ; WIEGHAUS, M. ; ROMMEL, M. (2003): Solar thermal-driven desalination plants based on membrane distillation. In: Elsevier Desalination : Volume 156 , 298-304. URL [Accessed: 07.03.2019] PDFDesalination by using Alternative Energy: Review of State of the Art
This paper presents a review of the highlights that have been achieved during the recent years and the state-of-the-art for most important efforts in the field of desalination by renewable energy, with emphasis on solar energy applications.
MATIOULAKIS, E. ; BELESSIOTIS, V. ; DELYANNIS, E. (2007): Desalination by using Alternative Energy: Review of State of the Art. In: Elsevier Desalination : Volume 203 , 346-365. URL [Accessed: 07.03.2019] PDFCalculating the Environmental Cost of Seawater Desalination in the Arabian Marginal Seas
In this paper, a mathematical model is presented to calculate the impact of desalination plants on salinity within a semi-enclosed sea of simple geometry.
PURNAMA, A. ; AL-BARWANI, H.H. ; SMITH, R. (2005): Calculating the Environmental Cost of Seawater Desalination in the Arabian Marginal Seas. In: Elsevier Desalination 185: Volume 185 , 79-86. URL [Accessed: 07.03.2019] PDFRenewable Energy for Desalination using Reverse Osmosis
This paper presents a proposal for fulfilling the energy demands of small desalination facilities in remote areas by using a combination of renewable energies. The integrated system consists of photovoltaic modules as well as a wind turbine, diesel generator, battery bank for energy storage and a reverse osmosis desalinisation unit.
SALAZAR, J. TADEO, F. PRADA, C. (2010): Renewable Energy for Desalination using Reverse Osmosis. Valladolid: Deparment of Systems Engineering and Automatic Control, University of Valladolid URL [Accessed: 05.03.2012]Dessalement de l’Eau de Mer: Bilan des Dernieres Avancés Technologiques; Bilan Economique; Analyse Critique en Fonction des Contextes
This article presents the state-of-the-art technologies in theworld desalination market. Modern techniques are described in detail and a critical comparison between different techniques is presented, taking into account economical and geographic criteria.
TATA-DUCRU, F. (2009): Dessalement de l’Eau de Mer: Bilan des Dernieres Avancés Technologiques; Bilan Economique; Analyse Critique en Fonction des Contextes. Paris: Agence Francaise de Developpement URL [Accessed: 07.03.2019] PDFLanguage: Spanish
Water Resources Management to Satisfy High Water Demand in the Aris Sharm El Sheikh, the Red Sea, Egypt
This paper describes the current water resource management practices in Sharm, and outlines simple strategies that could be undertaken to improve the situation.
LAMEI, A. ; ZAAG, P. van der ; MUNCH, E. von (2009): Water Resources Management to Satisfy High Water Demand in the Aris Sharm El Sheikh, the Red Sea, Egypt. In: Desalination and Water Treatment : Volume 1 , 299-306. URLPromotion of Solar Desalination in the MENA Region
Desalination has become one of the main sources of water in most of the countries in the Middle East and North Africa (MENA) region. All MENA countries lie in the Sunbelt region and have the space needed by solar technologies. Success in implementing solar technologies in desalination depends on the progress made or to be made to economically convert solar energy into electrical and thermal energies.
QUTEISHAT, K. ABU-ARABI, M. (2004): Promotion of Solar Desalination in the MENA Region. Muscat: Middle East Desalination Research Center URL [Accessed: 07.03.2019] PDFDesalination and San Francisco Bay
This case study provides recommendations for better management of desalination plants in San Francisco Bay. Environmental issues, operation, and technologies are also discussed.
SFBCDC (2005): Desalination and San Francisco Bay. San Francisco: San Francisco Bay Conservation and Development Commission (SFBCDC) PDFSolar Desalination: A Comparative Analysis
This poster presents a technical and economic comparison between main water desalination technologies.
TAILLEFER, Z. (n.y): Solar Desalination: A Comparative Analysis. Worcester: Worcester Polytechnic Institute URL [Accessed: 26.06.2012]International Desalination Association (IDA)
The International Desalination Association’s homepage announces events, showing publications, and linksinthis field.
Desalination - Directory Online
This is the International Desalination and Water Reuse Quarterly industry website, containing the latest news, products, and magazine articles.
http://www.desline.com/
This website presents publications, books courses etc.related to desalination.


